|
1/11/2024 0 Comments Shorthand notation anode cathode
We will have solid Zinc and we will have Copper 2+ ions, so we write that in here. So we add these together, and we know that the two electrons that were lost by Zinc are the same electrons thatĪre gained by Copper 2+, we can cancel those out, and so for our reactants 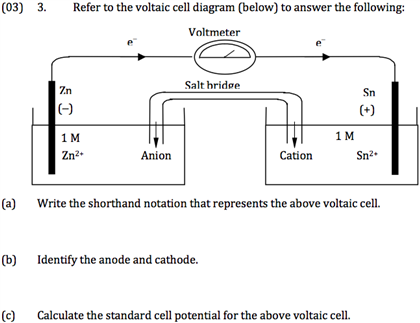
If we add together our two half-reactions we get our overall redox reaction. So "Leo the Lion Goes Ger", is a good way to remember So remember, loss ofĮlectrons is oxidation, and gain of electrons is reduction. So this would be solid Copper, and since we gained electrons this is our reduction half-reaction, so this represents a So think about what happens if you add two electrons to a Copper 2+ ion, you get solid Copper, right? So overall zero charge. And when those Copper 2+ ions come in contact with those electrons we get a reduction half-reaction. In solutions, this is an aqueous solution of Copper sulfate, so we have Copper 2+ ions in solution. So now we have these two electrons on our Copper electrode. So we have a wire set up and those two electrons are going to move, which is our electric current, right? So we get an electric current in our wire, and those two electrons move over to the electrode on the Those two electrons that we lost, these two electrons right here, are going to travel along our wire. So we've lost electrons, remember, when you lose electrons it's an oxidation reaction, or you could also lookĪt the oxidation states. This is our oxidation half-reaction, so this is an oxidation. So this is the losing two electrons, so we put the electrons So we have solid Zinc losing two electrons to turn into Zinc 2+ ions in solution. 
The atom will have to lose two electrons, so two electrons are leftīehind on the Zinc electrode. 
And so, we already know what happens on this electrode on the left, the Zinc electrode or the solid Zinc turns into Zinc 2+ ions. Remember, a Voltaic Cell uses a spontaneous redox reaction to create an electric current. Here is an example for reference.Get into shorthand notation let's review the structure of the Galvanic or Voltaic Cell. Also it is important to note that aqueous solutions should be directly next to the double lines. On the very outermost right, we have the substance that is reduced, and to the left of that single line, we see what it looks like before it is reduced. The very left of this cell diagram we have the substance that gets oxidized (for instance Zn(s)) and then on the right side of the single vertical line is what the substance looks like once it is oxidized and loses its electrons. On the left side of the double lines, you have the anode (where the oxidation takes place) and on the right side you have the cathode (where the reduction takes place). The single lines represent the phase change of the substance and the double lines represent the salt bridge that the electrons are flowing on towards the other side. With the short hand notation, it is called a "cell diagram", and we are basically representing what is occurring when you have a redox reaction. The only thing that matters in terms of sides is that the right side is the species in the cathode and the left side is the species in the anode. In lecture, Dr Lavelle also said that for the purpose of Chem 14B, it doesn't matter what order you put your specific species in on one of the sides, so I believe that Cu (s) | Cu 2+ (aq) and Cu 2+ (aq) | Cu (s) would both be accepted answers. Use a comma if they are the same phase (i.e. if there is a conducting metal in the solution like Pt). Cu (s) | Cu 2+ (aq)) or if they are different species (i.e. To separate species in one side of the reaction (cathode OR anode), you would use a single line if they are in different phases (i.e. The single line represents the interface between two species in different phases in contact with each other, but it can also be used to represent a porous disk. For a reaction using a porous disk, you can use a single line between the species present in the anode and the species in the cathode. For a cell diagram with a salt bridge, you would use a double line in between the species present in the anode and the species in the cathode to indicate the presence of the salt bridge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
